-

“If the compound can be made, Symeres will undoubtedly find out a good way to make it.” Dooyoung Jung, CEO of Pinotbio.
An in-depth interview with Doo Young Jung, the founder and CEO of our client Pinotbio in South Korea, focused on the challenge of cancer resistance/relapse
-

New video of our Solid-State Center of Excellence
In this new video our colleagues Gabriella Pizutti, Chantalle van Berkel, and Eva Verhofstad, will give you an exciting lab tour through the Symeres Solid-State Centre of Excellence, located at the Symeres site in Weert, the Netherlands. You will learn more about PhysChem Properties, Solid Form Screening, Crystallization, Crystal Habit, Pre-formulation, and Formulation. Feel free to contact us via the contact form on this website if you would like to learn more about our Solid-State capabilities.
-
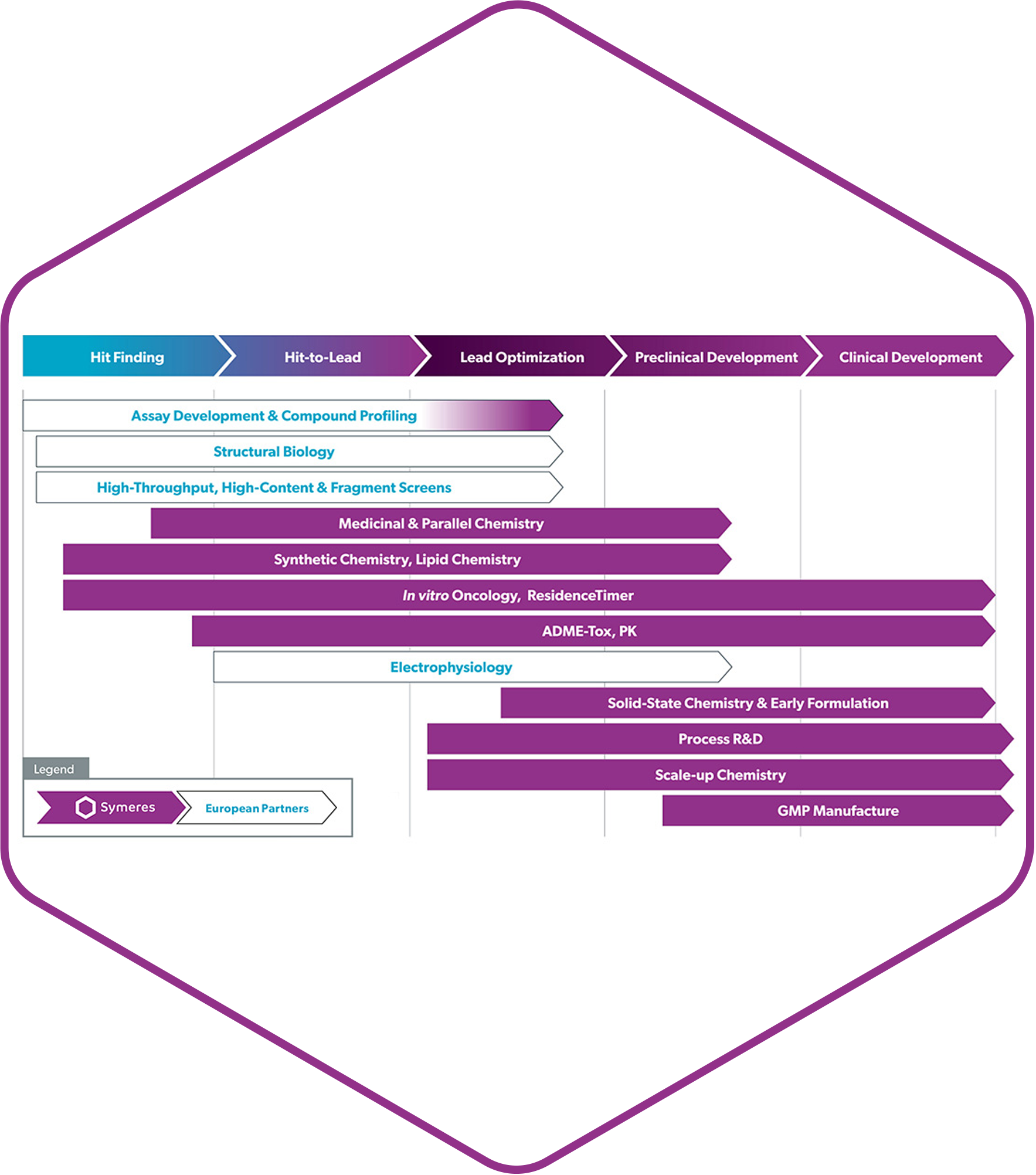
New scientific services from Symeres
We’re proud to announce that our brand new CMC facility at the Exemplify site of Symeres in New Jersey has been successfully completed. GMP drug product R&D facilities that will allow it to provide end-to-end services ranging from GMP and non-GMP drug product development to phase 1 and 2 clinical supplies of various oral, parenteral, and topical formulation dosage forms.
-

Interview with the new Managing Director of Symeres Groningen
On October 2, Dr Melloney Dröge started in her new role as Managing Director for the Groningen site.
-
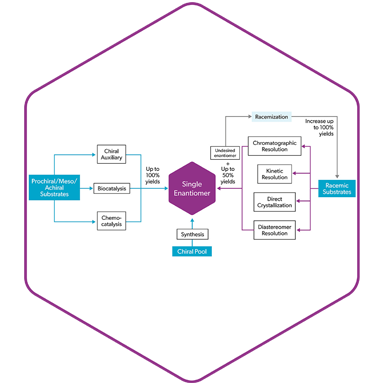
Chiral Chemistry: in the DNA of Symeres
Over 50% of current drugs in development are chiral. At Symeres we apply a suite of state-of-the-art technologies to obtain the desired chiral compound in high enantiomeric purity. For decades, Symeres has been supporting the global pharma and biotech industry with the synthesis and delivery of chiral compounds, both through synthesis and purification strategies.
-

Interview with Richard Blaauw, Director of Operations at Organix
In April 2022, Symeres acquired Organix, a US-based CRO specializing in lipid synthesis. To aid in the incorporation of Organix into the Symeres family, and facilitate future growth transitions, Richard Blaauw has been appointed to the newly created position of Director of Operations.
-

Symeres Weert receives new GMP certificate
This week, our facility in Weert NL received a new certificate of GMP compliance from the Dutch authorities.
-

A decade of integrated drug discovery at Symeres
Symeres celebrates a decade of integrated drug discovery. Our team has been working hard to provide our clients with the best possible solutions for their needs.
-

Symeres welcomes Karen Damian to the team
We introduce you to our newest Director of Business Development. Karen Damian has a history in the drug development life sciences business.
-
π-Facial selectivity in the Diels–Alder reaction of glucosamine-based chiral furans and maleimides
Symeres explorations into opportunities to access novel chemical space by using carbohydrates as sustainable chiral starting materials for library synthesis has led to yet another publication.
-

An interview with Yadan Chen and Paul O’Shea
We are pleased to introduce the founders of Symeres’ daughter company Exemplify in New Jersey: Yadan Chen, CEO, and Paul O’Shea, Chief Scientific Officer. Who are they? What do they stand for? And how does Exemplify fit with Symeres?
-

We’re expanding our laboratory facilities in Boston
To continue supporting our clients, our daughter company Organix needs more room. The new facility will help us expand our services.
Feel free to contact us!
Curious to know more about what can we do for you? Get in touch and let’s start a conversation.
